Zeolite Anammox De-Ammonification Process
Collison Engineering
WHAT IS THE ZEOLITE ANAMMOX PROCESS?
The Zeolite-anammox process is designed to remove ammonia and reduce Total Nitrogen (TN). By removing nitrogen in its reduced form, this treatment process prevents the formation of nitrate.
ZEOLITE
-
Zeolite is a natural mineral in a rock matrix, with high cation exchange capacity
-
The rock selected for zeolite-anammox reactors preferentially removes ammonia
-
The zeolite cation exchange sites adsorb the positively charged ammonium ions
-
The Western USA is fortunate in having massive zeolite deposits in California, Nevada, & New Mexico

ANAMMOX
-
Anammox was discovered 20 years ago and has revised the nitrogen cycle
-
Anammox bacteria derive their energy from eating NH4
-
Half the ammonium NH4+ coming from wastewater gets oxidized to nitrite NO2-. The anammox combines the NO2- with the remaining NH4+ to directly form nitrogen gas N2
-
NH4 + NO2 = N2 + 2H2O
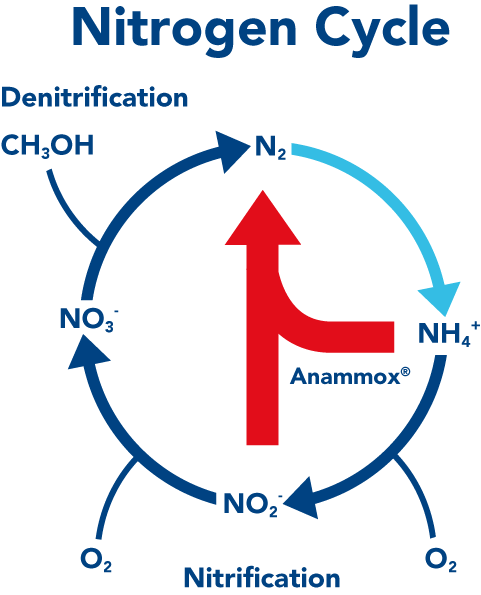
In the presence of anammox the left hand side of the nitrogen cycle (including nitrate and denitrification) does not occur to significant degree.
ZEOLITE-ANAMMOX
1) IMMOBILIZATION
The zeolite-anammox process uses zeolite to immobilize ammonium by cation exchange (CEC).
2) COLONIZATION
Bacteria find ammonium-coated zeolite very attractive, and colonize it. A biofilm forms attaching the bacteria to the zeolite.
3) REGENERATION
Bacteria living on the zeolite oxidize half the ammonium to nitrite. Anammox bacteria combine ammonium and nitrite to form nitrogen gas and water. Since the bacteria eat the NH4+ and regenerate the zeolite in-situ it never becomes saturated.
4) SINGLE STAGE
Overall, the process is single stage as the anammox take NH4+ and convert it directly to nitrogen gas.

Advantages of Zeolite-Anammox Compared to Traditional Denitrification
-
Requires half the infrastructure
-
Uses half the air
-
Significant cost savings
Flat Bed Reactors

-
Small flows up to 3MGD
-
Horizontal flow by gravity – no pumping, agitation or aeration needed
-
Low energy requirements (aeration & pumping typically form 80% of a city’s public works energy budget per CWEA)
-
The system is simple; the infrastructure is economical; the operating cost is low
-
Proven NH4 removal: 300mg/L → 0.1mg/L
Aerated Reactors

-
Larger flows >3MGD
-
Aeration added to supplement oxidation and increase efficiency → smaller footprint
-
Aerobic/anaerobic or oxic/anoxic zones for symbiotic nitrifiers and anammox
-
Cheaper than alternatives (nitri/deni), but more expensive than flat bed option